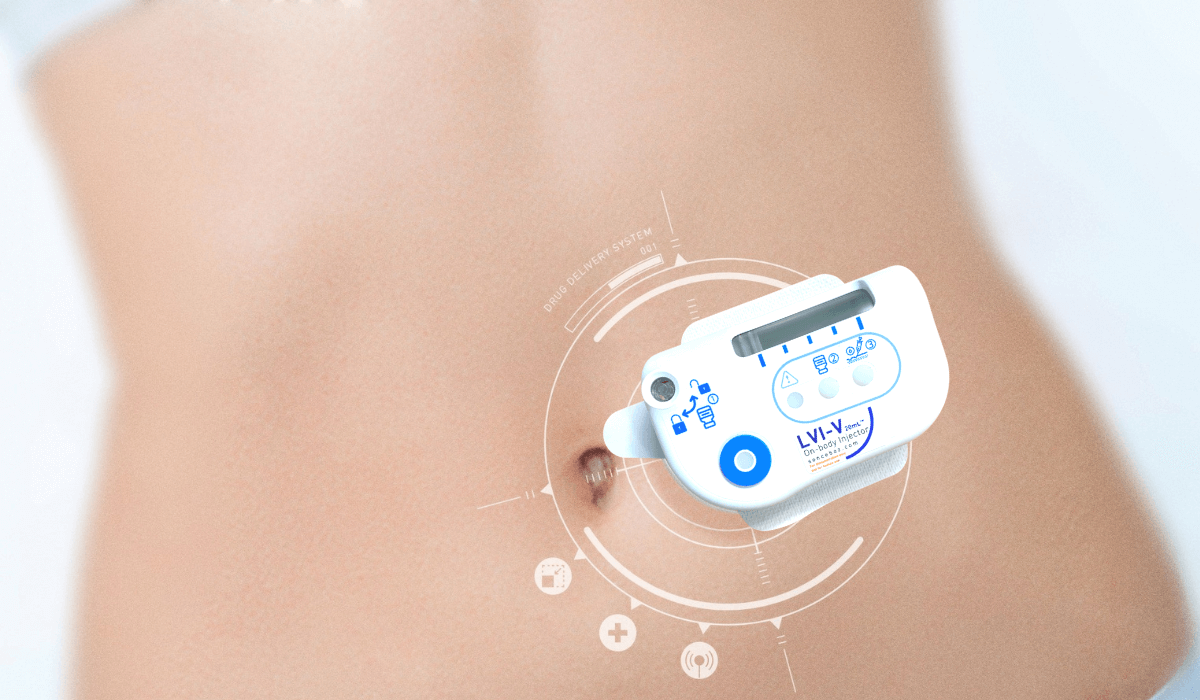
Being active at work, or enjoying dinner with friends while receiving medication continuously, discreetly and comfortably: this is a dream for many patients. Sonceboz technology makes this dream a reality by offering innovative ways to fight chronic diseases. Our wearable drug delivery systems dispense high volumes of viscous medications thereby offering patients the freedom they desire.



THERAPEUTIC MEMBERSHIP
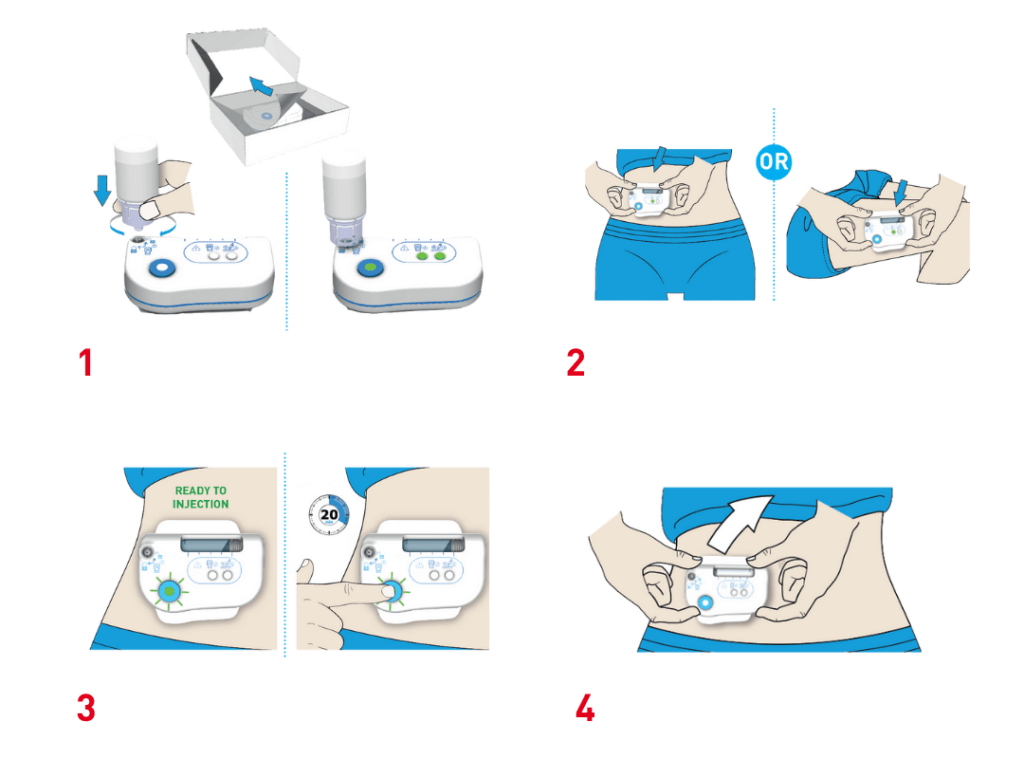
STEPS OF USE
DEVICE PLATFORM
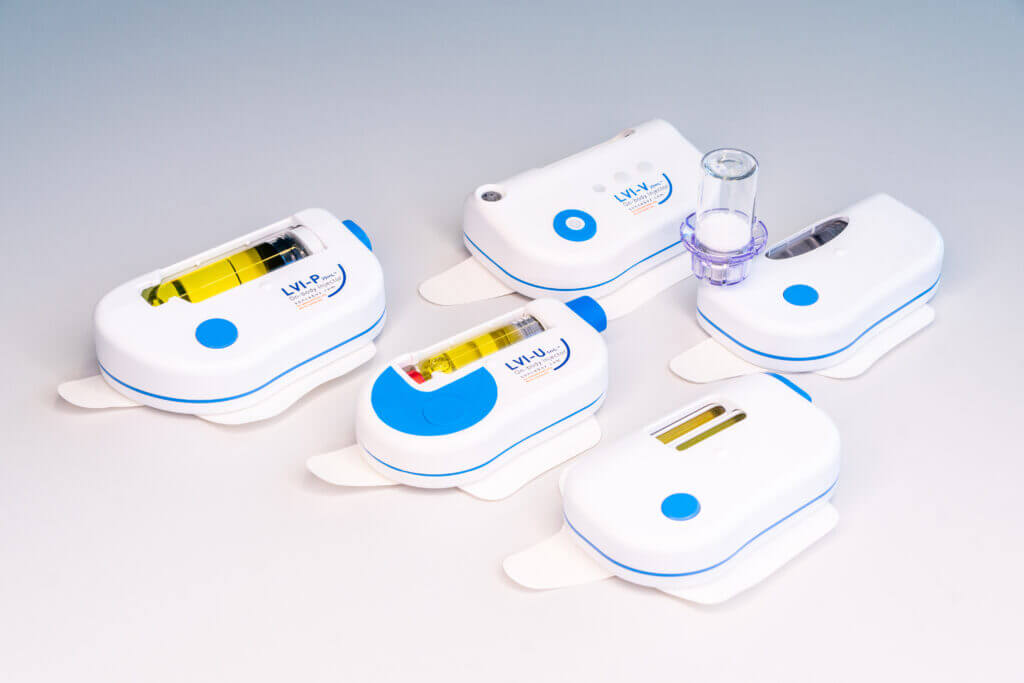
The unique wearable injection device platform accompanying drugs from clinical trials to commercial phase and life cycle management – in top Swiss quality at world market prices.
Faster time to market
- Versatile platform offering unprecedented therapeutic options from single container to dual container and automatic reconstitution while using 90% identical parts
- Easy customization and branding
- Full container flexibility from vials to cartridges. This allows for utilization of existing fill and finish solutions.
Less risk
- GentleTouch™ piston pump for enhanced drug compatibility
- Highly reliable and proven brushless drive technology
- Soft cannula with automatic needle insertion and retraction offering patient comfort and preventing needle-stick injuries
Quality and cost
- Complete service from device design, customization, validation and manufacturing
- High integration of device components for best cost, quality and reliability
- High degree of automation
- Developed according to ISO11608
Technical Data
- Drug container volumes between 1 and 20mL
- Flow rate 0 to 1,5ml/min
- Viscosity up to 100cp
- Minimal dosing increment 50nl
- Dosing precision +/- 4%
- Residual system volume < 0,03ml
- Low noise level < 35dBA
- 27G soft cannula
- Connectivity with Bluetooth-LE
APPLICATION AREA
LVI-V™
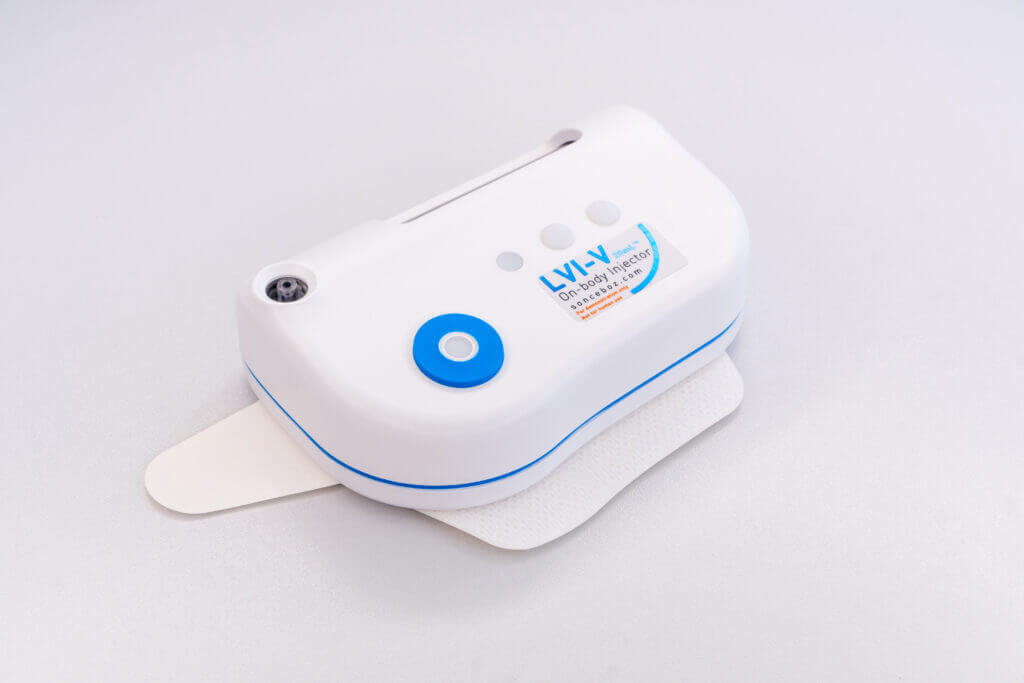
- Clinical readiness: 2024
- Compatible to standard vials up to 20mL
- Fast to clinic
- Ideal for (remote) trial use or HCP-Assisted use
LVI-P™

- Clinical readiness: 2025
- Tailored for self-injection
- Prefilled and Preloaded with up to 20mL
- Focus on intuitive and easy use
- Bridging from LVI-V
CUSTOMER SPECIFIC DEVELOPMENTS
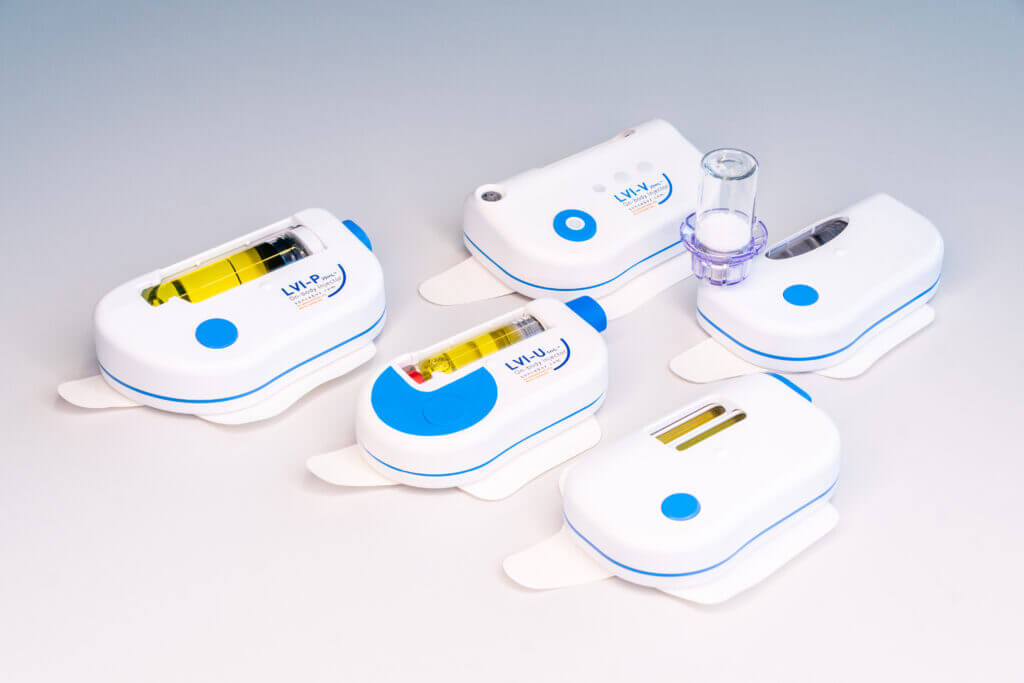
- High degree of flexibility
- Large range of delivery volumes 3-20mL
- Common technology platform
- Differentiating : Multi-Day, Dual-Cartrige use or Automatic Reconstitution
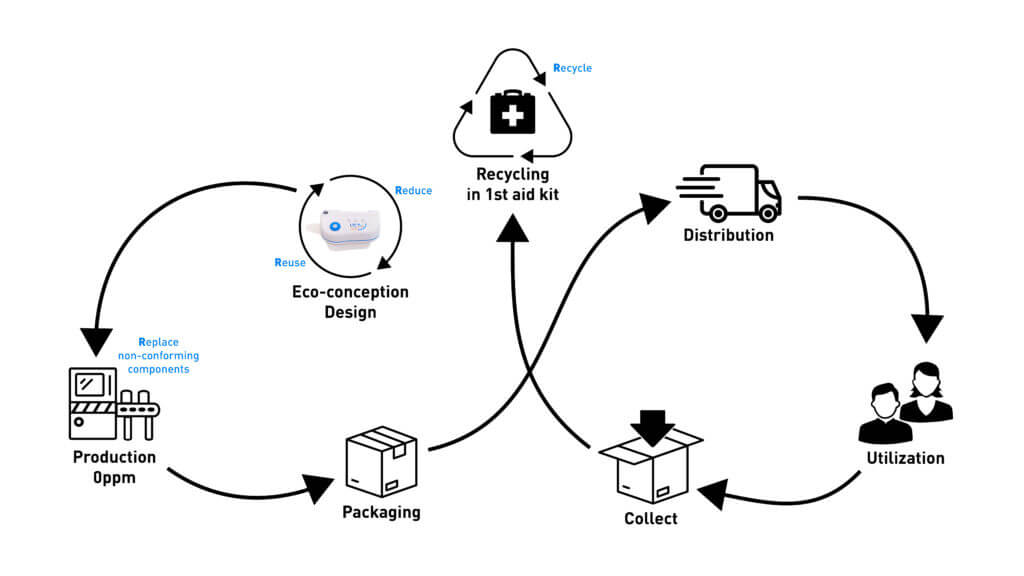
Sustainability
Sonceboz applies whenever possible the 4R-model (Reduce, Reuse, Replace, Recycle) during the entire lifecycle of its On-body injector (OBI) device, especially during design and manufacturing:
- Reduce as much as possible the carbon footprint
- Reuse as much as possible the same material for several components
- Replace and scrap non-conforming components by checking every single part throughout the assembly process for 0ppm defect
- To recycle by creating an efficient collection network and sorting components from used devices
Because recyclable does not mean recycled, Sonceboz is actively working on the end-of-life of its OBI and the possible second life for the materials used. Indeed, sustainability is an effort to reduce the impact on the patient and the environment, keeping in mind that reusing parts of disposable devices is challenging due to the validation process. As safety cannot be compromised, recycling components into other medical devices might not be feasible. However, transforming parts into other tools such as first-aid kits may be a very interesting alternative that Sonceboz is investigating closely.
Additionally, since the OBI will enable home treatment thereby reducing patient travel, it will dramatically reduce the impact on the environment while minimizing the risks of contamination, for example with hospital acquired infections.